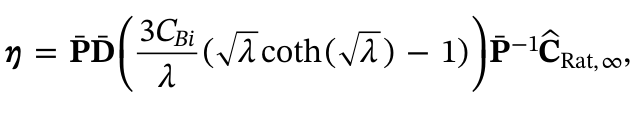MultiStage Effectiveness Vector (MEV)
Chemical transformation by heterogeneous catalysts is critical to processes involving hydrogenation, isomerization, hydration, oxidation, syngas upgrading, and petroleum refining operations. Improvements to current catalytic chemical technologies, from environmental as well as economic perspectives, are guided by process intensification, integration, and optimization approaches. An important tool that is central to all of these approaches has been, and will continue to be, multiphase reaction engineering.
Though common in many industrial chemical processes, multi-step reactions occurring within porous catalyst particles provide significant challenges to previously established modeling frameworks. In this work, a system of reaction-diffusion equations is solved analytically via eigendecomposition. The intraparticle concentration profiles are integrated over the particle volume to obtain effectiveness factors for each reacting species, referred to herein as a multi-step effectiveness vector (MEV):
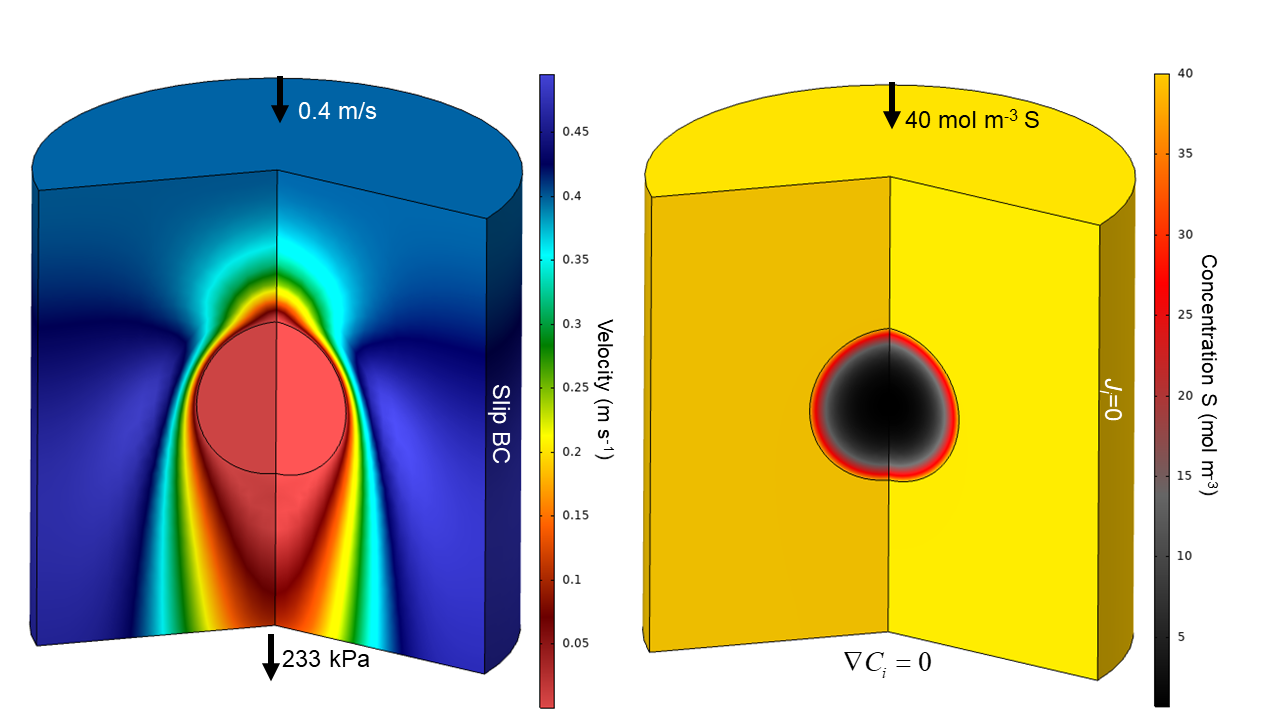
The MEV solution marks a significant advancement of the classic single-stage effectiveness factor (R. Aris, “The mathematical theory of diffusion and reaction in permeable catalysts: The theory of the steady-state,” Oxford University Press, 1975), which is only valid for a simple A -> B type reaction. Considering a state-of-the-art, multi-step reaction scheme for fluid catalytic cracking (FCC) and uniform flow past a stationary catalyst particle:
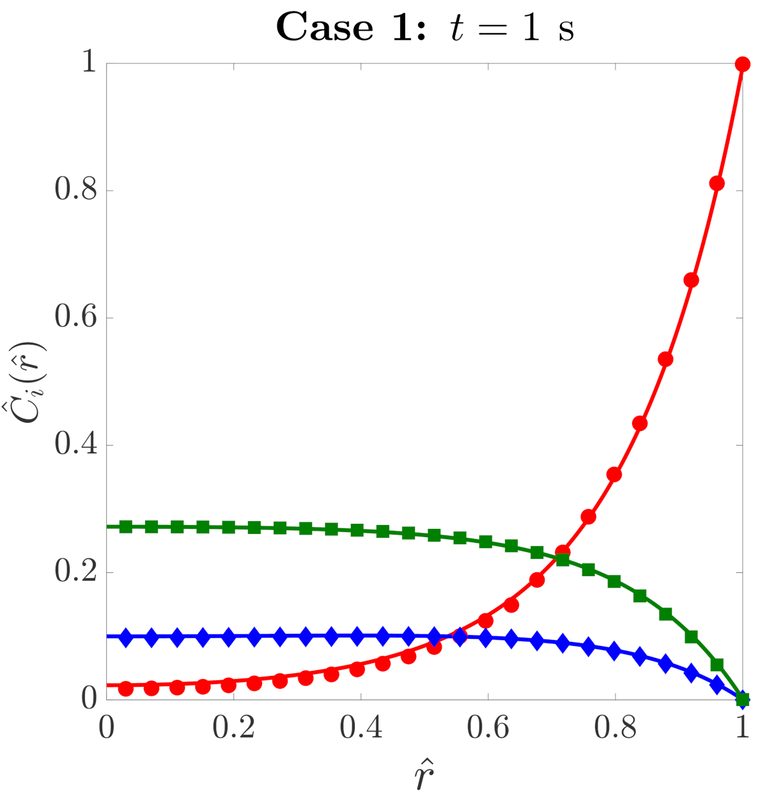
the MEV shows excellent agreement with concentration profiles obtained from fully-resolved simulations:
The MEV theory is being actively developed into a computational library for linking to flow solvers and currently supports sphere, cylinder, and prism geometries:
- Lattanzi A.M., Pecha M., Bharadwaj V. & Ciesielski P. 2020 Beyond the effectiveness factor: Multi-step reactions with intraparticle diffusion limitations. Chemical Engineering Journal, 380, 122507.